Pankaj Patel's Inspirational Journey: From Cadila Laboratories To Owner Of Rs 52,400 Crore Empire
Raised in a traditional Patel family, Patel studied at Sheth C. N. Vidyalaya and later earned a Master of Pharmacy degree from L M Pharmacy, Ahmedabad.
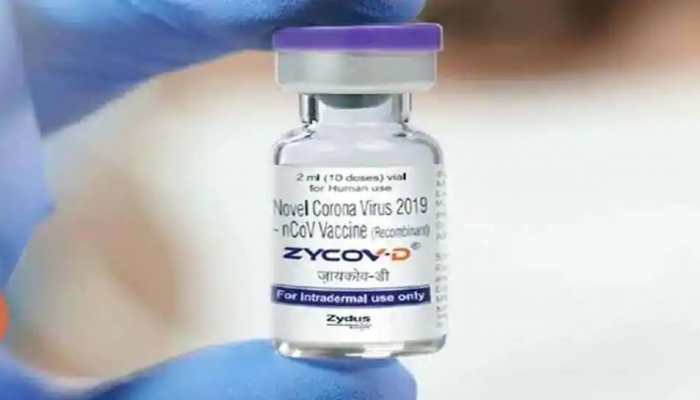
Dec 31, 2023, 10:22 AM ISTAll about ZyCov D, India's first needleless vaccine launched in Bihar's Patna
ZyCov D is a plasmid DNA vaccine that uses a genetically engineered, non-replicating version of a type of DNA molecule known as a ‘plasmid.'
Feb 05, 2022, 09:51 AM ISTZydus Cadila's COVID-19 vaccine ZyCoV-D to be initially introduced in THESE seven states
Union Health Secretary Rajesh Bhushan directed these states to identify the districts with a high number of beneficiaries who have not taken even the first dose of the COVID-19 vaccine.
Dec 02, 2021, 16:06 PM ISTCentre places order for one crore doses of Zydus Cadila's COVID vaccine ZyCoV-D
The Union Health Ministry is learnt to have given the go ahead to initiate the preparatory work for the introduction of the indigenously developed ZyCoV-D.
Nov 07, 2021, 14:25 PM ISTZydus Cadila to reduce COVID vaccine price to Rs 265, final decision soon
Zydus Cadila's COVID-19 vaccine is the first vaccine to be approved by India's drug regulator for inoculation to 12 years and over.
Oct 31, 2021, 18:20 PM ISTCOVID19: Biological E gets nod for phase 2/3 clinical trial for vaccine on children
Biological E has received approval for conducting phase II/III human clinical trial of COVID-19 vaccine candidate Corbevax on children above five years and adolescents.
Sep 03, 2021, 16:33 PM ISTPriority COVID-19 vaccination for children above 12 with comorbidities: Govt panel chief
NTAGI chief said that a portion of Zydus Cadila’s Zycov-D vaccine supply will be set aside for comorbid adolescents. The announcement came days after the Drug Controller General of India (DCGI) granted emergency approval to the Zycov-D.
Aug 24, 2021, 07:35 AM ISTZydus Cadila hopes to begin supply of ZyCoV-D vaccine by mid to end September, says MD Sharvil Patel
Patel also said that the pricing of the ZyCoV-D dose will be announced in the next one or two weeks.
Aug 22, 2021, 07:25 AM ISTZydus Cadila's ZyCoV-D - the needle-free COVID-19 vaccine for adults and kids - all you need to know
Zydus Cadila in July had said that ZyCoV-D was effective against the new mutants of COVID-19, especially the Delta variant.
Aug 21, 2021, 10:22 AM ISTZydus Cadila's COVID vaccine ZyCoV-D, first jab for children in India, gets approval
Zydus Cadila Covid vaccine ZyCoV-D can be administered to people 12 years and above.
Aug 20, 2021, 20:57 PM ISTZydus Cadila likely to get emergency use approval for its COVID vaccine this week: Sources
Zydus Cadila had said last month that it has applied for emergency use authorisation (EUA) with the Indian Drug regulator for its three-dose COVID-19 vaccine ZyCoV-D, and plans to manufacture 10-12 crore doses annually.
Aug 09, 2021, 21:30 PM ISTZydus Cadila seeks DCGI's permission for emergency use of its COVID-19 vaccine for those above 12
Gujarat-based pharmaceutical company Zydus Cadila has completed the Phase III trial of its coronavirus vaccine. Following which, the company has sought approval from the Drugs Controller General of India (DCGI) for emergency use of this drug on people above 12 years of age.
Jul 01, 2021, 11:24 AM ISTZydus Cadila seeks emergency use approval for COVID vaccine for 12 years old and above
The company said that it has applied to the country`s drug regulator for emergency use approval of its COVID-19 vaccine and that it plans to manufacture up to 120 million doses of the shot annually.
Jul 01, 2021, 09:09 AM ISTCOVID-19 vaccine Zydus Cadila for children above 12 expected by August, says NTAGI chief
Zydus Cadila COVID-19 vaccine for children above 12 years is expected to be available by August as its trials are likely to be completed by July-end, said Dr N K Arora, Chairman, National Technical Advisory Group on Immunisation (NTAGI).
Jun 27, 2021, 18:02 PM ISTCan 'Virafin' be a viable alternative for Remdesivir as COVID-19 wreaks havoc in India
The price of the drug is yet to be decided but it will be positioned competitively against Remdesivir
Apr 24, 2021, 13:39 PM ISTDNA: Virafin, a hope to eradicate COVID-19
Zydus Cadila on Friday received approval from the Drug Controller General of India (DGCI) for emergency use of the drug Virafin. Antiviral Virafin will reportedly make treatment more convenient for COVID-19 patients.
Apr 23, 2021, 22:00 PM ISTEmergency use of Zydus Cadila's drug Virafin approved against COVID-19
To speed up the treatment of Coronavirus patients, the Drug Controller General of India (DGCI) on Friday approved the emergency use of Zydus Cadila's drug Virafin. This medicine can be used to treat patients above 18 years of age.
Apr 23, 2021, 17:02 PM ISTBreaking news: DGCI approves emergency use of Zydus Cadila's Virafin for moderate COVID-19 infection
Drugs Controller General of India, DGCI, on Friday (April 23) approved the emergency use for Zydus Cadila's Pegylated Interferon alpha-2b, ‘Virafin’ for treating moderate COVID-19 infection in adults.
Apr 23, 2021, 15:53 PM ISTWe will have an amazing New Year Today, After Vaccination approval?
The approval for a Covid vaccine can come soon, the country's drugs control authority has indicated.
Jan 01, 2021, 12:06 PM ISTCorona Vaccine : Pfizer Seeks India Approval For Covid Vaccine
Pfizer has sought approval from the country's drug regulator - the DCGI (Drugs Controller General of India) - for emergency use authorization of its coronavirus vaccine.
Dec 06, 2020, 14:12 PM IST